HSR Initial and Amendments Portal
Human Subject Research Portal
Before you start, you will need:
- IRB (COMIRB) Tracking Number – to obtain a COMIRB number (24-XXXX), select CU Denver from the InfoEd eRA login page. Then, click Human Protocol, and Create New Human Protocol, do not click submit.
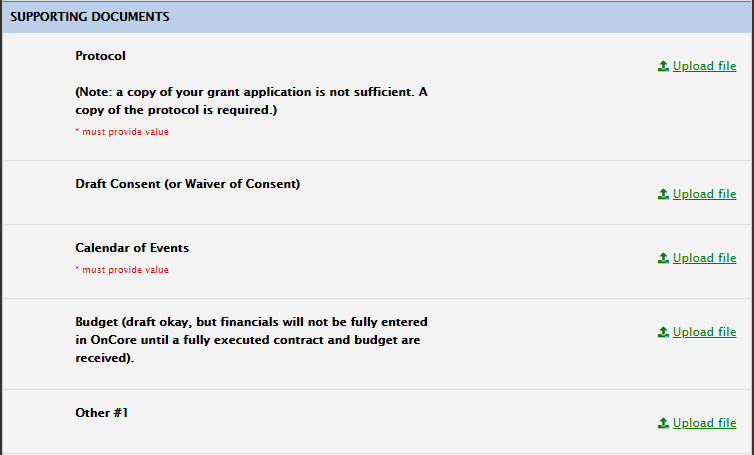
- Protocol (draft is acceptable)
- Consent (for studies that have patient interaction; draft is acceptable)
- Draft contract (if industry funded/industry initiated)
- Calendar of Events (draft is acceptable)
- You’ll be able to include other optional documents to help us review your study.
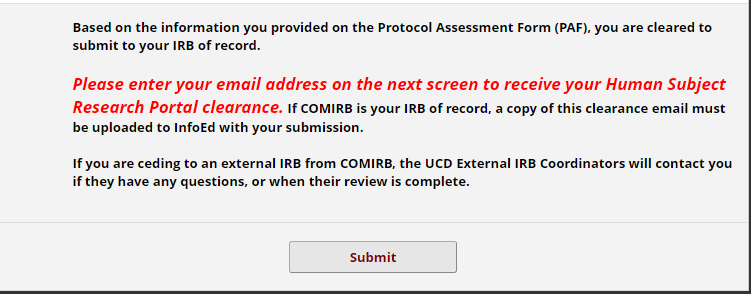
IMPORTANT: COMIRB requires a Portal Clearance Memo with your submission for initial review of non-exempt research.
Enter your email address when prompted, and you’ll receive the memo from [email protected] once the review is complete.
**Please make sure to check your Inbox and save this email as Clinical Research Administration will not be copied.**
CHCO only studies: Portal Clearance Memos not required; provide your CHCO approval letter to COMIRB instead.
If you have received a Just-In-Time (JIT) notification from the NIH for IRB approval, submit your study to COMIRB first. Submit your study materials to the HSR Portal promptly after COMIRB approval.
If your study is investigator initiated, locally written with no previous scientific review, it will require either PRMS (if oncology) or SARC (all other non-cancer studies) review to receive portal clearance.
For Children’s Hospital Colorado studies:
You must complete the CHCO ancillary form, as part of the portal submission process. COMIRB does not require Portal Clearance Memos for CHCO only studies. You will provide them your CHCO approval letter instead.
Need to save and come back to your submission?
You have the option to save and return later if you start the form and need to some back. Just write down your return code!
You can finish your submission later on if you are not ready by clicking Save & Return Later at the bottom of the submission page:

This will provide a return link and code. Please only use this if you have not fully submitted your portal form. If you need to return and make corrections to an already submitted study, please request the code and link from OnCore Support ([email protected]).
Using the return code and link, you will be given two options after you click the link:
- Submit a return code
- Start over
If the “start over” button is selected, it will remove all responses from the form. Please use the option only when you need to delete every entry from your portal survey.
NOTE: Submissions started but not completed within 90 days will be deleted.
Study Information Section

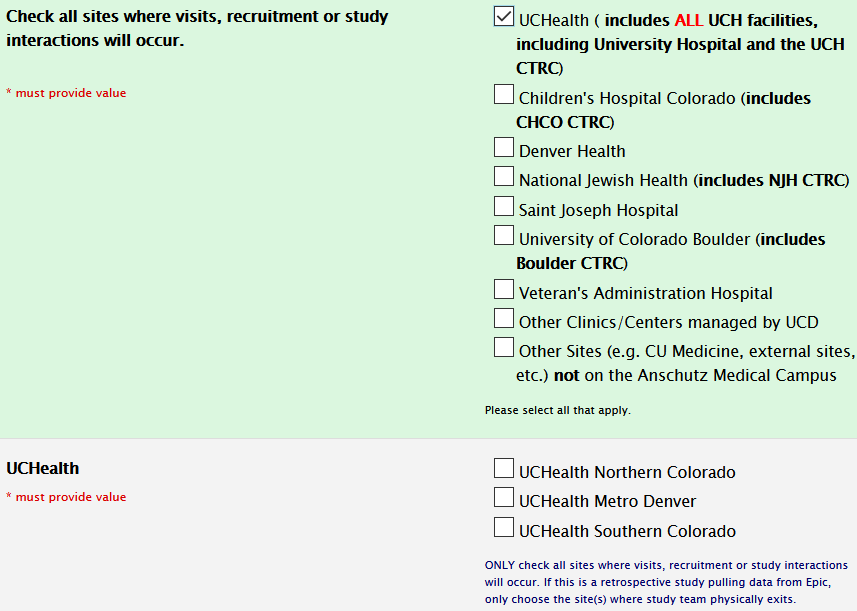
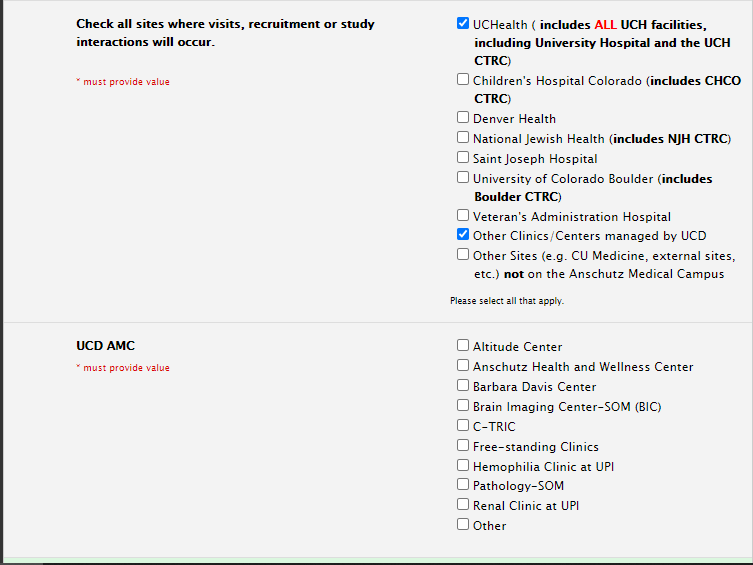
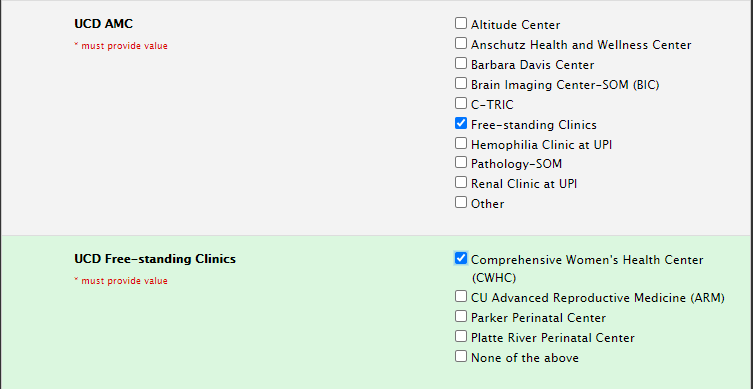
Study Information Section: Sites
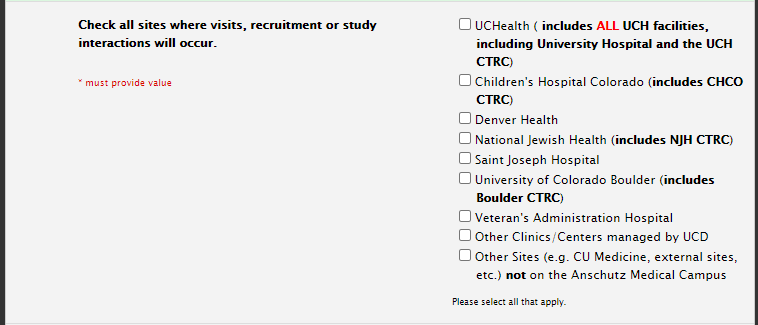
Sites: Retrospective Studies
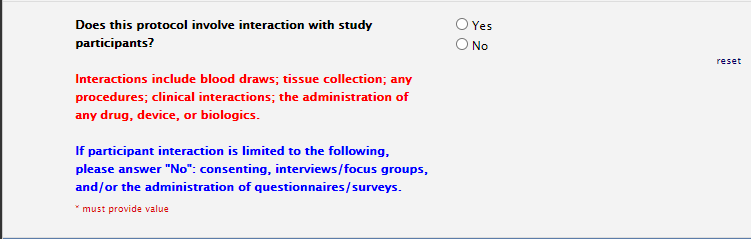
Study Information Section: Patient Interaction
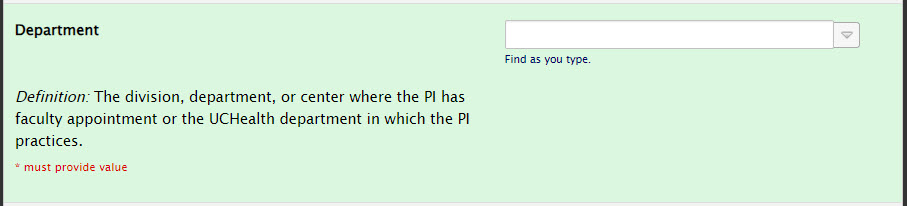
Principal Investigator Section: Department
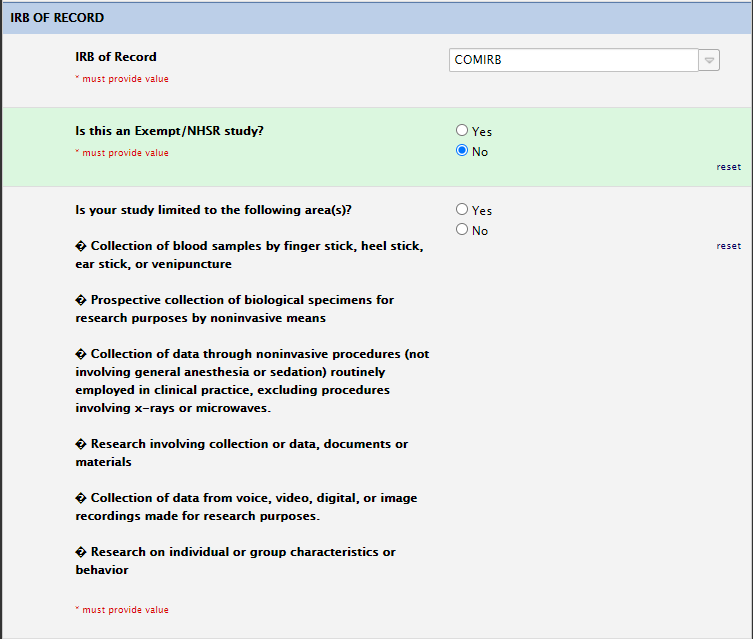
IRB of Record Section
Staff Section
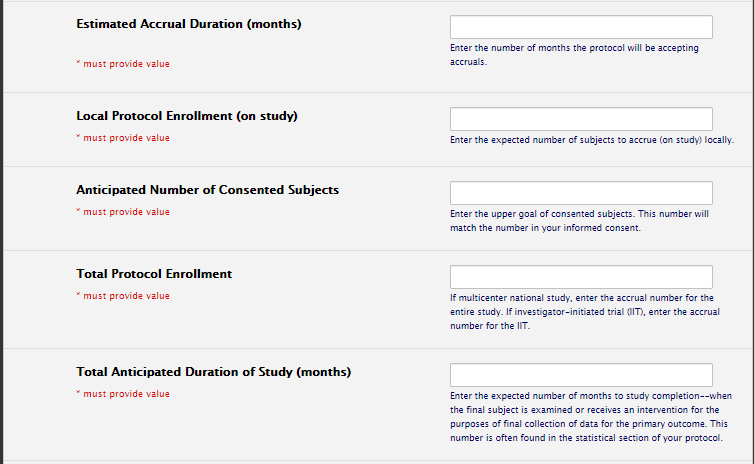
Calendar Section
Feasibility/Recruitment Plan Section
Supporting Documents Section
Submission: Final Steps
Selecting Sites
Selecting Sites: Free-standing Clinics
Oncology
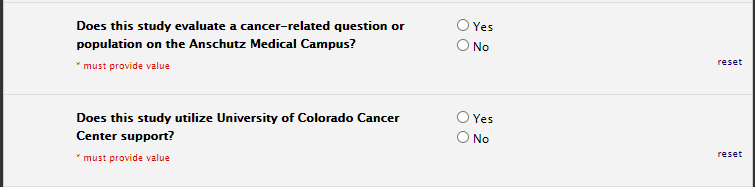
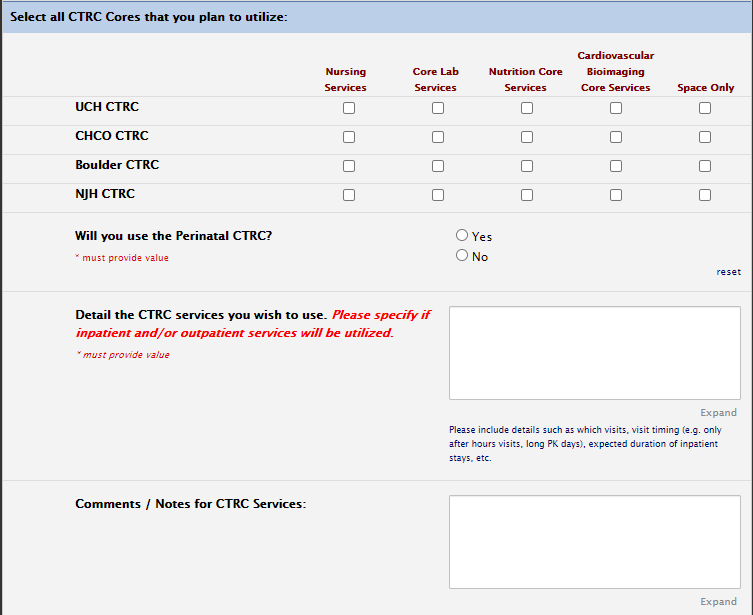
Funding Section: CTRC
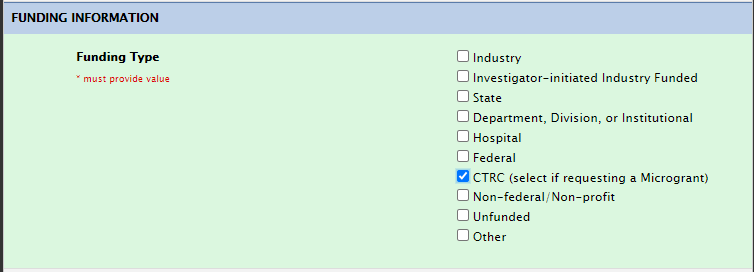
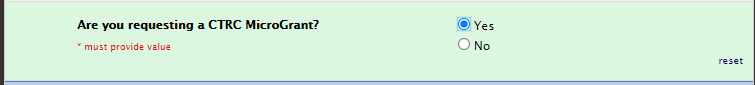
CTRC MicroGrant
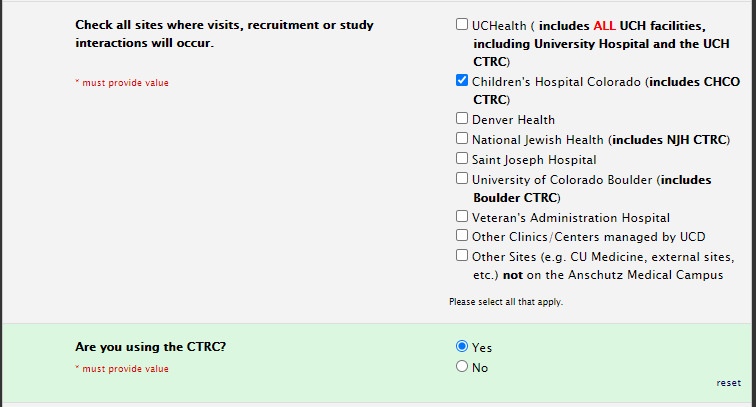
Site selection for CTRC usage
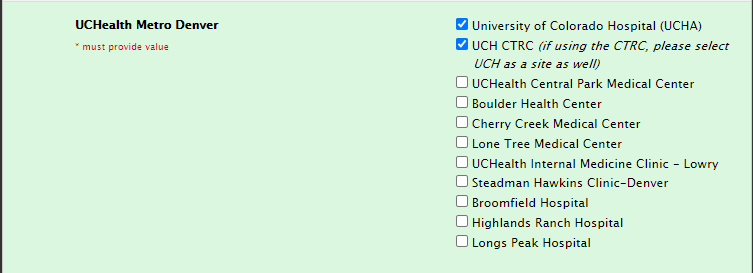
CHCO CTRC
CTRC services
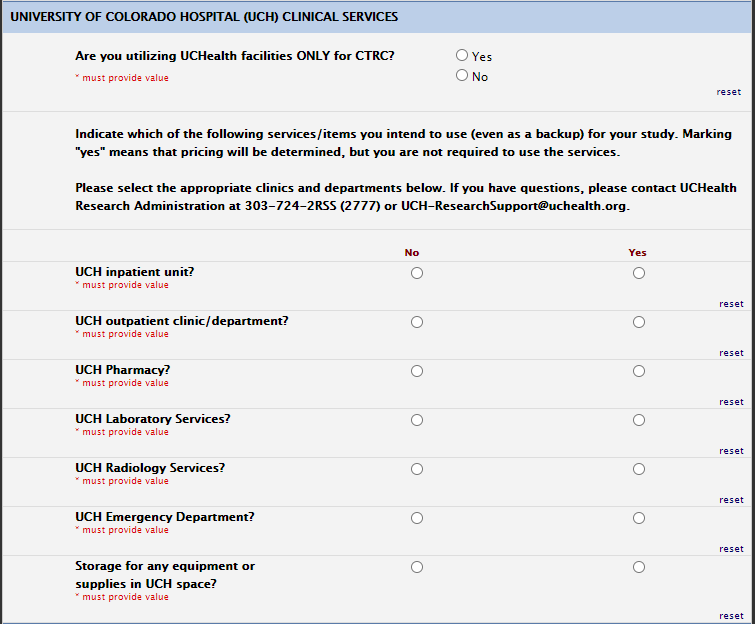
UCH Services:
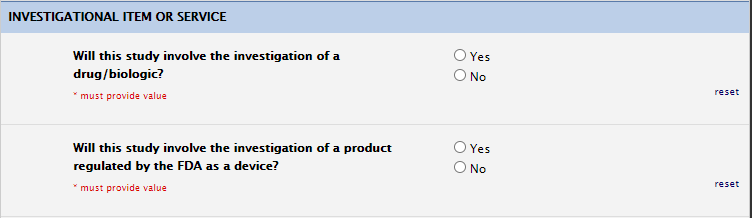
Investigational Item or Service
After you submit to the Amendment Portal, Clinical Research Administration and OnCore Support teams review the changes for regulatory and billing compliance implications.
Information for Sponsors
The University of Colorado has developed a partially executed CDA (Confidential Disclosure Agreement).
If your sponsor can accept this document by including legal address, effective date and signing, the CDA will be fully executed and may be used. We have instructed our study teams how to work with you on the use of this document.
This partially executed CDA will decrease completion time for a fully executed CDA.